Physics Notes On – Amorphous Structure Of Matter – For W.B.C.S. Examination.
Physics is an optional subject for W.B.C.S. Mains Exam. It is a discipline of science which deals with properties and nature of matter and energy. It includes light, heat, mechanics and other related themes. Just go through the W.B.C.S. Main optional Physics syllabus and you will find that aspirants those who are from Physics and Electrical engineering background can handle this optional subject. W.B.C.S. aspirants coming from non-electronics background must put extra hard work and endeavor to ace this optional.Amorphous solids are rigid structures but they lack a well-defined shape. They do not have a geometric shape. So they are non-crystalline. This is why they do not have edges like crystals do. The most common example of an amorphous solid is Glass. Gels, plastics, various polymers, wax, thin films are also good examples of amorphous solids.Continue Reading Physics Notes On – Amorphous Structure Of Matter – For W.B.C.S. Examination.
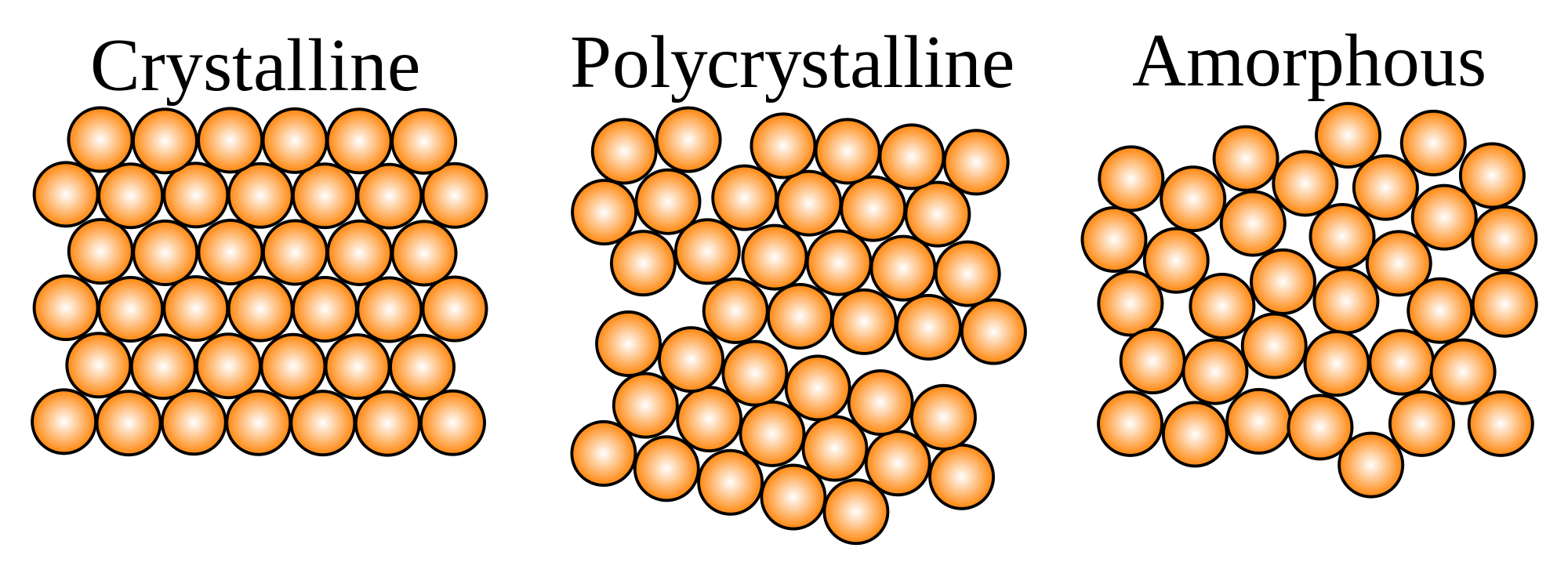
This variation in characteristics of solids occurs due to the arrangement of their molecules. Here the particles of matter do not form the three-dimensional lattice structure that we see in solids. Some naturally occurring amorphous solids have impurities that prevent such a structure from forming. So they have a short order arrangement of molecules.
Amorphous solids break into uneven pieces with irregular edges. And they do not have any distinct arrangement or shape of molecules. so they cannot be identified by their structure as crystals.
Amorphous solid, any noncrystalline solid in which the atoms and molecules are not organized in a definite lattice pattern. Such solids include glass, plastic, and gel.
Solids and liquids are both forms of condensed matter; both are composed of atoms in close proximity to each other. But their properties are, of course, enormously different. While a solid material has both a well-defined volume and a well-defined shape, a liquid has a well-defined volume but a shape that depends on the shape of the container. Stated differently, a solid exhibits resistance to shear stress while a liquid does not. Externally applied forces can twist or bend or distort a solid’s shape, but (provided the forces have not exceeded the solid’s elastic limit) it “springs back” to its original shape when the forces are removed. A liquid flows under the action of an external force; it does not hold its shape. These macroscopic characteristics constitute the essential distinctions: a liquid flows, lacks a definite shape (though its volume is definite), and cannot withstand a shear stress; a solid does not flow, has a definite shape, and exhibits elastic stiffness against shear stress.Also Read , Motivational Books To Read – Motivation And Inspiration – For W.B.C.S. Examination.
On an atomic level, these macroscopic distinctions arise from a basic difference in the nature of the atomic motion. contains schematic representations of atomic movements in a liquid and a solid. Atoms in a solid are not mobile. Each atom stays close to one point in space, although the atom is not stationary but instead oscillates rapidly about this fixed point (the higher the temperature, the faster it oscillates). The fixed point can be viewed as a time-averaged centre of gravity of the rapidly jiggling atom. The spatial arrangement of these fixed points constitutes the solid’s durable atomic-scale structure. In contrast, a liquid possesses no enduring arrangement of atoms. Atoms in a liquid are mobile and continually wander throughout the material.
Please subscribe here to get all future updates on this post/page/category/website


 +919674493673
+919674493673  mailus@wbcsmadeeasy.in
mailus@wbcsmadeeasy.in